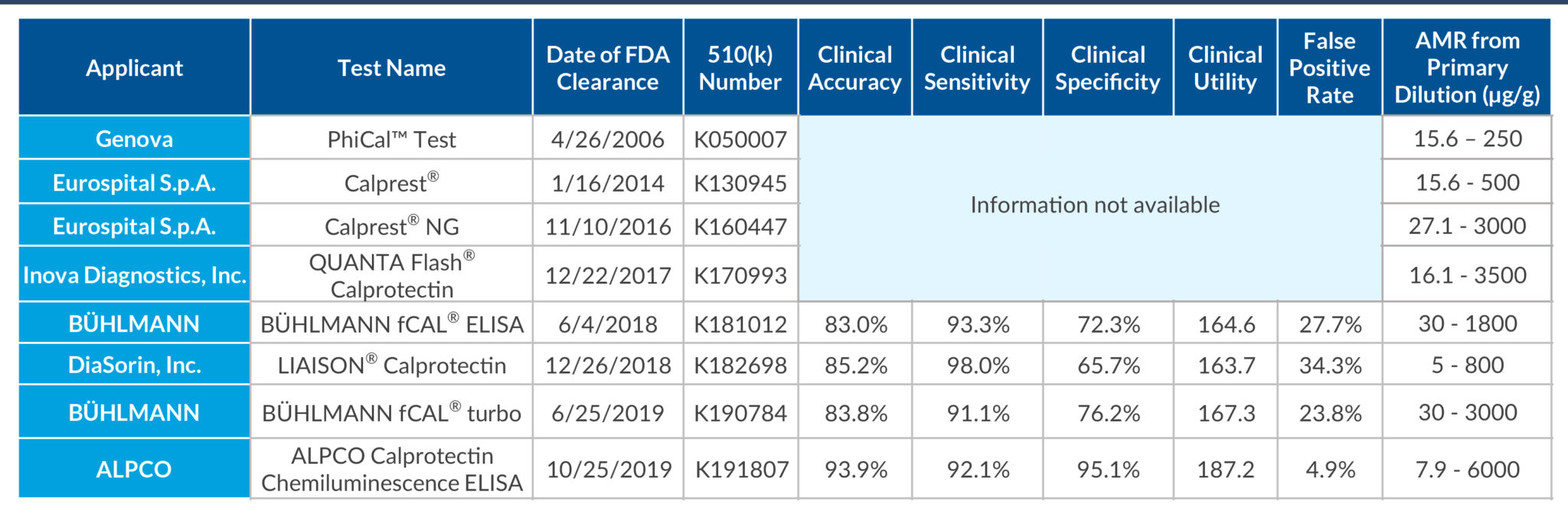
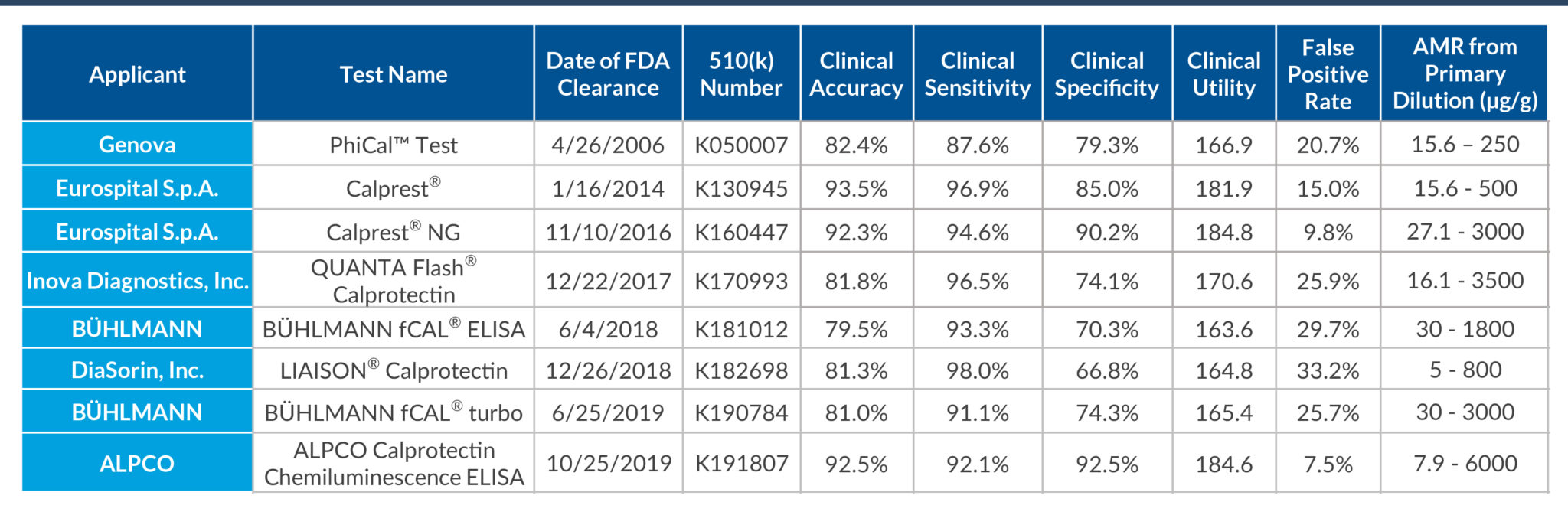
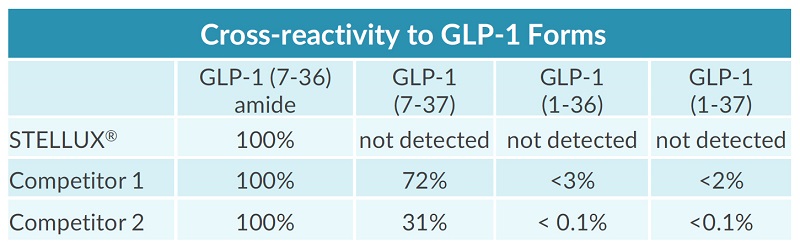
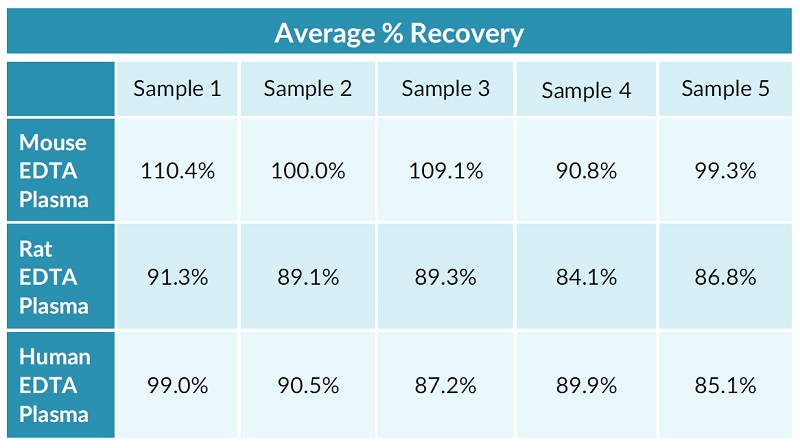
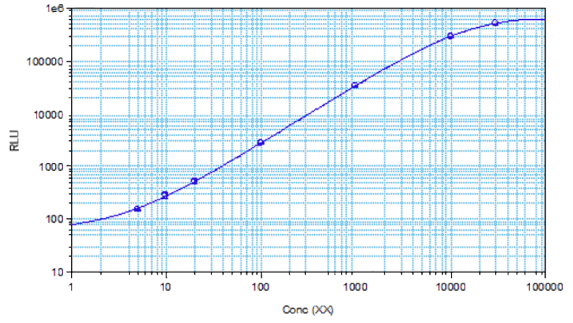
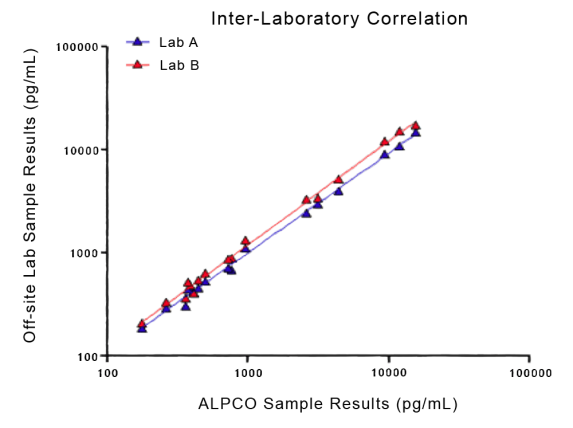
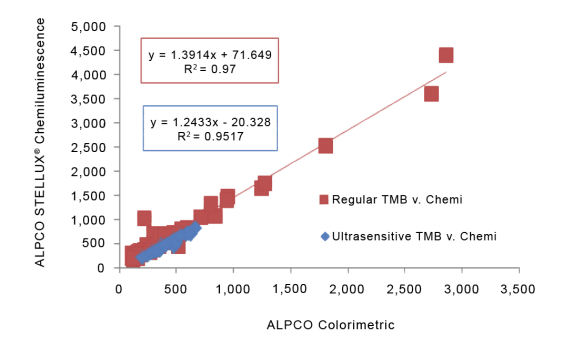
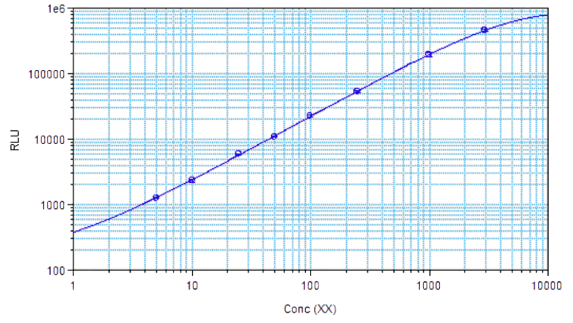
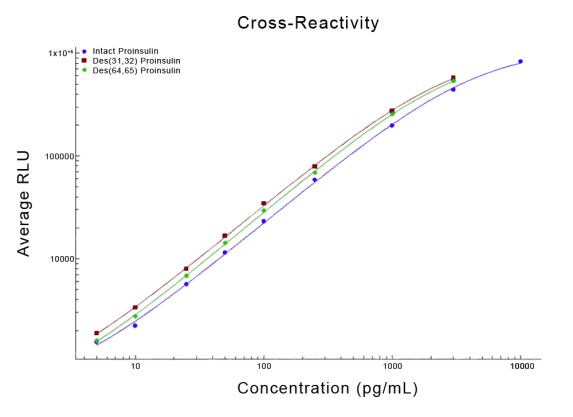
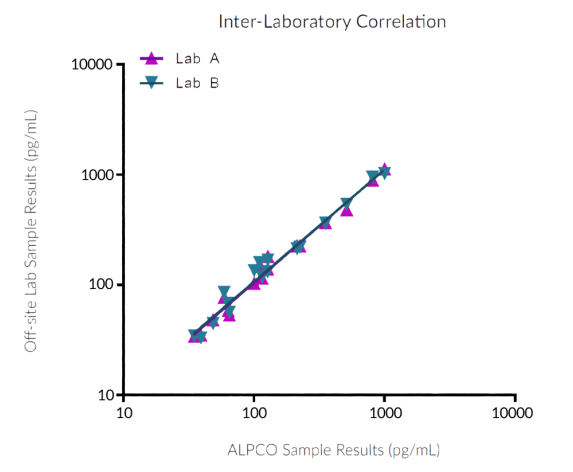
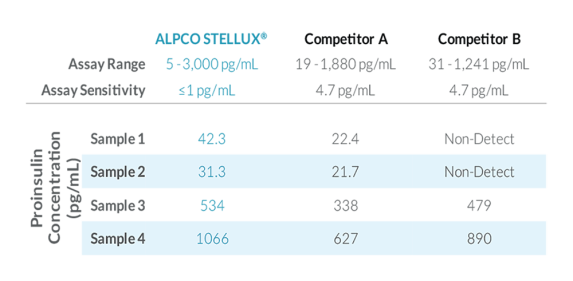
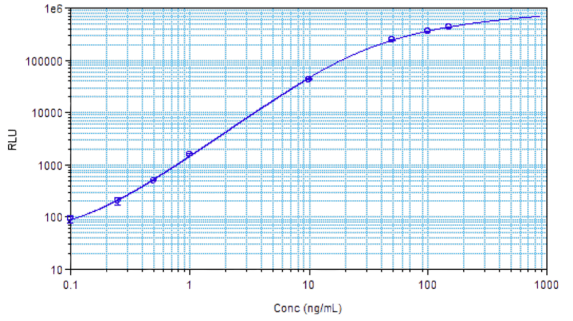
STELLUX® is our chemiluminescence ELISA platform aimed at detecting key biomarkers in life science research.

Alpco diagnostics are a supplier of immunoassay kits for the medical research community.
Their kits are developed by scientists committed to supporting those who work in physiology and disease research.
ALPCO provide a broad portfolio of life science research tools for academic, pre-clinical and clinical use, including primary antibodies, flow cytometry reagents, recombinant proteins, and products for HPLC and LC-MS/MS, but are best known for providing, as they put it: “Immunoassays Beyond the Ordinary.”
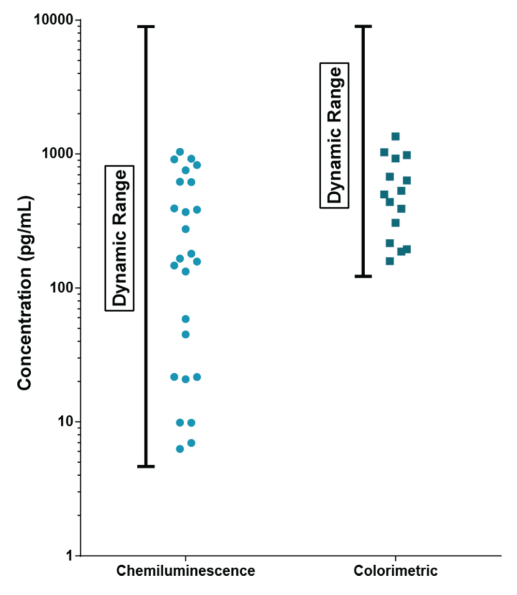
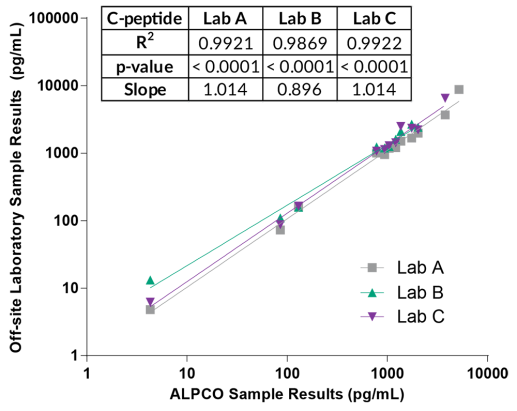
ALPCO have a reputation for quality among their customers because they focus on providing immunoassays which are highly sensitive, specific and reproducible with a broad dynamic range that allows for fewer dilution steps and therefore reduced systematic error.
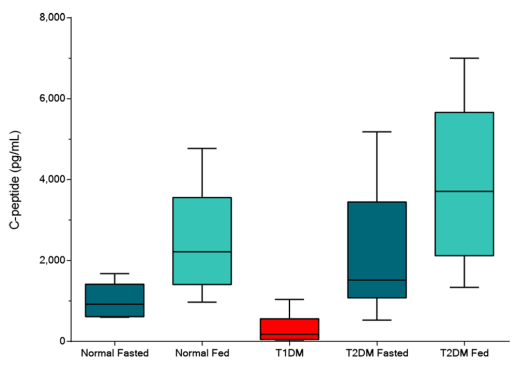
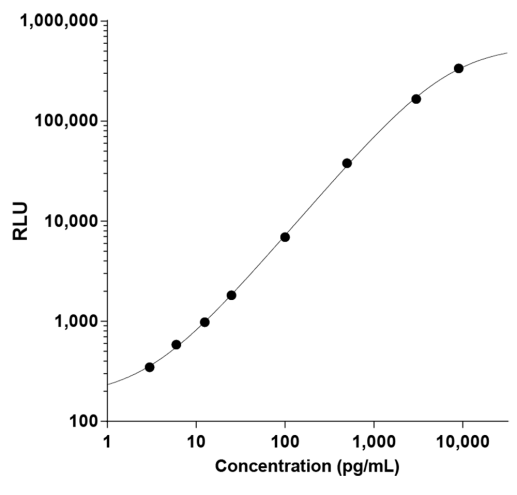
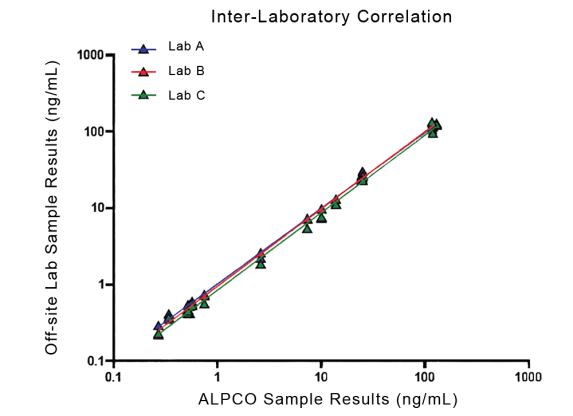
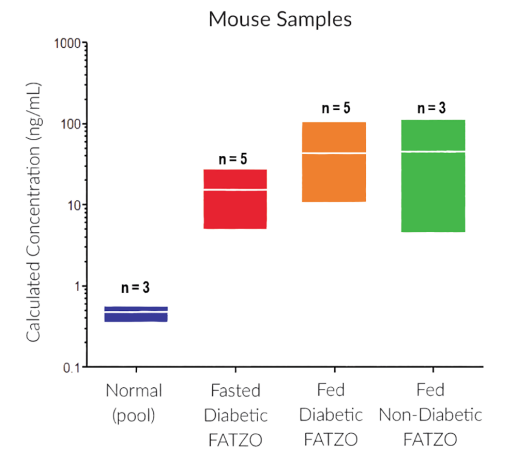
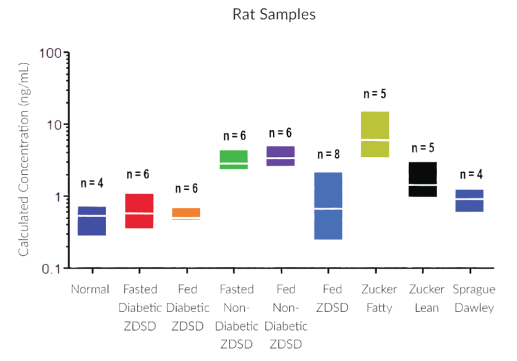
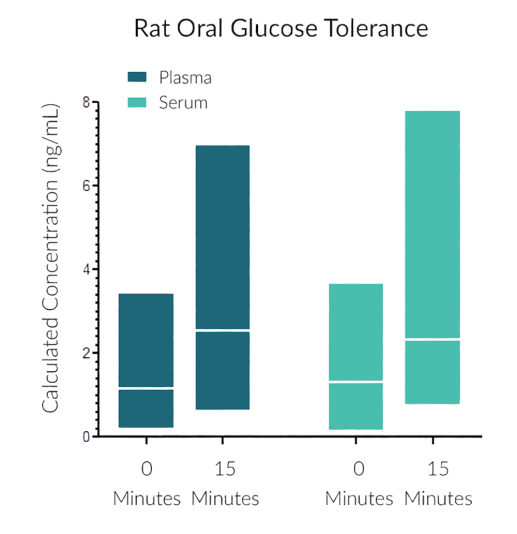
They have recently developed the new STELLUX™ range of chemiluminescent immunosorbent assays. These combine exquisite sensitivity, ease of use, and outstanding reliability which comes from extensive testing and characterisation.