Research on SARS-CoV-2 is currently a key concern of life sciences to prevent further infections and to develop a vaccine. The following studies as well as the antigen specific FluoroSpot method demonstrate the great utility of the Strep-tag® technology in this area. It can be used for the isolation of cells and exosomes or for affinity purification of proteins, but moreover Strep-Tactin®XT proves to be particularly good for the immobilization and detection of viral proteins due to its high affinity for the Twin-Strep-tag®.
Research tools for Covid-19
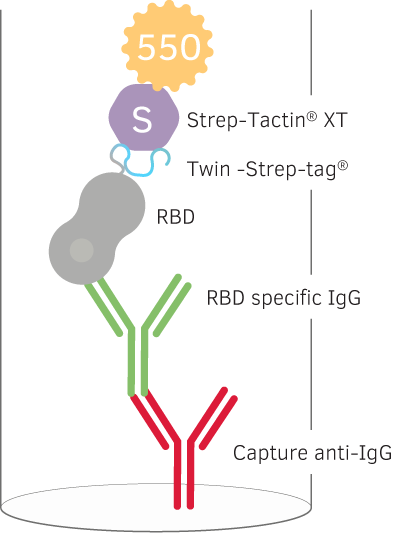
Detection of Memory B cells specific for SARS-CoV-2 with reversed antigen specific IgG FluoroSpot
The Swedish biotech company Mabtech showed that reversed antigen specific FluoroSpot is a valid method for testing human B cell memory response, which could be an interesting complement for studying long term immunity of COVID-19.
In their study they immobilized their anti-human IgG (MT91/145) capture antibodies in a FluoroSpot plate. For detection of IgGs specific to a receptor binding protein (RBD) of the SARS-CoV-2 Spike protein, the RBD was fused with a C-terminal Twin-Strep-tag® that subsequently was detected with a Strep-Tactin® XT conjugated to a 550-fluorophore. They decided to recombinantly express the Twin-Strep-tag® together with the RBD protein instead of traditional biotinylation, because biotinylation of lysines might interfere with immunogenic epitopes.
More relevant papers for Covid-19 research
Gordon et al., made great progress in finding therapies that may reduce the mortality of COVID-19. The researchers identified the human proteins that associate with viral proteins of SARS-CoV-2 using an affinity-purification mass spectrometry approach. In this regard, they expressed Twin-Strep-tagged versions of SARS-CoV-2 proteins in human cells, which they could verify by anti-Strep western blot. Next, Gordon and colleagues purified the expressed Twin-Strep-tagged viral proteins together with associated human proteins using MagStrep “type3” XT beads. After elution of the viral proteins with buffer BXT, they analyzed the co-eluted proteins by mass spectrometry and could reveal 332 human proteins that with high confidence interact with SARS-CoV-2. Out of these proteins, 66 can be targeted by existing drugs that are either approved, in clinical or pre-clinical stages. In a first screen for their anti-viral properties, two sets of pharmacological agents seem promising, inhibitors of mRNA translation and predicted regulators of the Sigma1 and Sigma2 receptors. Although further studies of the potential therapeutics for efficacy in SARS-CoV-2 infection are required, the run towards a COVID-19 treatment might be accelerated.
Our tools for COVID-19 research
Monoclonal antibody conjugated to DY-649 to detect Strep-tag®II and Twin-Strep-tag® fusion proteins
Conjugated monoclonal antibody to detect Strep-tag®II and Twin-Strep-tag® fusion proteins

Strep-Tactin® AP conjugate for detection of Strep-tag®II and Twin-Strep-tag® fusion proteins

Strep-Tactin® conjugate for detection of Strep-tag®II and Twin-Strep-tag® fusion proteins
Strep-Tactin®XT conjugate with fluorescent label to detect Strep-tag®II and Twin-Strep-tag® fusion proteins
Strep-Tactin®XT conjugated to fluorescent DY-549 to detect Strep-tag®II and Twin-Strep-tag® fusion proteins
Strep-Tactin®XT conjugated to fluorescent DY-649 to detect Strep-tag®II and Twin-Strep-tag® fusion proteins
Monoclonal antibody conjugated to DY-549 to detect Strep-tag®II and Twin-Strep-tag® fusion proteins
Conjugated monoclonal antibody to detect Strep-tag®II and Twin-Strep-tag® fusion proteins

Monoclonal antibody conjugated to DY-649 to detect Strep-tag®II and Twin-Strep-tag® fusion proteins

Conjugated monoclonal antibody to detect Strep-tag®II and Twin-Strep-tag® fusion proteins
Low affinity Fab fragment fused to a Twin-Strep-tag® and specific for human CD8
Gravity flow column suitable for traceless affinity cell and exosome isolation
MHC class I molecule fused to a Twin-Strep-tag® and refolded with a peptide

50% suspension to purify Strep-tag®II or Twin-Strep-tag® fusion proteins
Magnetic beads (5% suspension) to purify Strep-tag®II or Twin-Strep-tag® fusion proteins
50% suspension to purify Strep-tag®II or Twin-Strep-tag® fusion proteins
Reversible immobilization of biotinylated analytes or Strep-tag®II and Twin-Strep-tag® fusion proteins