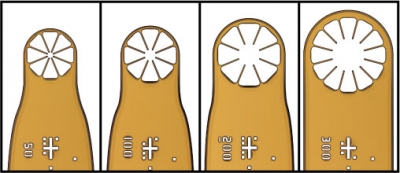
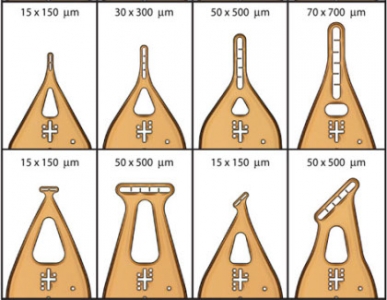
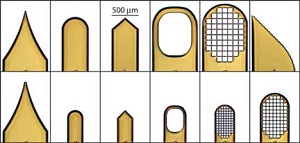
| Dual-Thickness MicroMount Assembly |
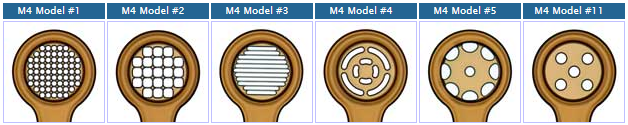
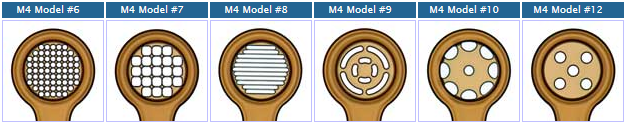
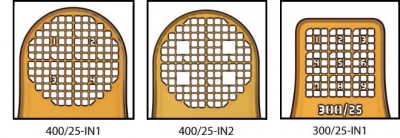
MicroMesh Assembly |
Dual-Thickness MicroLoop Assembly |
Dual-Thickness MicroLoop LD Assembly |
MicroLoop E Assembly |
MicroGripper Assembly |
| Crystal Mount-Base-Vial-Assembly HTA-M2-XX-BY Dual Thickness MicroMount Assembly with specified aperture size (xx) on 18mm/SPINE length rods adhered into specified barcoded bases (By) with cryovials |
Crystal Mount-Base-Vial-Assembly HTA-M3-XX-BY MicroMesh Assembly with specified aperture size (xx) on 18mm/SPINE length rods adhered into specified barcoded bases (By) with cryovials |
Crystal Mount-Base-Vial-Assembly HTA-M5-XX-BY MicroLoop Assembly with specified aperture size (xx) on 18mm/SPINE length rods adhered into specified barcoded bases (By) with cryovials |
Crystal Mount-Base-Vial-Assembly HTA-M5-XXLD-BY MicroLoop LD Assembly with specified aperture size (xx) on 18mm/SPINE length rods adhered into specified barcoded bases (By) with cryovials |
Crystal Mount-Base-Vial-Assembly HTA-M8-XX-BY MicroLoop E Assembly with specified aperture size (xx) on 18mm/SPINE length rods adhered into specified barcoded bases (By) with cryovials |
Crystal Mount-Base-Vial-Assembly HTA-M7-XX-BY MicroGripper Assembly with specified aperture size (xx) on 18mm/SPINE length rods adhered into specified barcoded bases (By) with cryovials |
| Crystal Mount-Base-Vial-Assembly HTA-M2-XX-BY-R Dual Thickness MicroMount Assembly with specified aperture size (xx) on 18mm/SPINE length rods adhered into specified barcoded reusable bases (By) with cryovials |
Crystal Mount-Base-Vial-Assembly HTA-M3-XX-BY-R MicroMesh Assembly with specified aperture size (xx) on 18mm/SPINE length rods adhered into specified barcoded reusable bases (By) with cryovials |
Crystal Mount-Base-Vial-Assembly HTA-M5-XX-BY-R MicroLoop Assembly with specified aperture size (xx) on 18mm/ SPINE length rods adhered into specified barcoded reusable bases (By) with cryovials |
Crystal Mount-Base-Vial-Assembly HTA-M5-XXLD-BY-R MicroLoop LD Assembly with specified aperture size (xx) on 18mm/ SPINE length rods adhered into specified barcoded reusable bases (By) with cryovials |
Crystal Mount-Base-Vial-Assembly HTA-M8-XX-BY-R MicroLoop E Assembly with specified aperture size (xx) on 18mm/SPINE length rods adhered into specified barcoded reusable bases (By) with cryovials |
Crystal Mount-Base-Vial-Assembly HTA-M7-XX-BY-R MicroGripper Assembly with specified aperture size (xx) on 18mm/SPINE length rods adhered into specified barcoded reusable bases (By) with cryovials |
| Crystal Mount-Base-Assembly HTB-M2-XX-BY Dual Thickness MicroMount Assembly with specified aperture size (xx) on 18mm/SPINE length rods adhered into specified barcoded bases (By) without cryovials |
Crystal Mount-Base-Assembly HTB-M3-XX-BY MicroMesh Assembly with specified aperture size (xx) on 18mm/SPINE length rods adhered into specified barcoded bases (By) without cryovials |
Crystal Mount-Base-Assembly HTB-M5-XX-BY MicroLoop Assembly with specified aperture size (xx) on 18mm/SPINE length rods adhered into specified barcoded bases (By) without cryovials |
Crystal Mount-Base-Assembly HTB-M5-XXLD-BY MicroLoop LD Assembly with specified aperture size (xx) on 18mm/SPINE length rods adhered into specified barcoded bases (By) without cryovials |
Crystal Mount-Base-Assembly HTB-M8-XX-BY MicroLoop E Assembly with specified aperture size (xx) on 18mm/SPINE length rods adhered into specified barcoded bases (By) without cryovials |
Crystal Mount-Base-Assembly HTB-M7-XX-BY MicroGripper Assembly with specified aperture size (xx) on 18mm/SPINE length rods adhered into specified barcoded bases (By) without cryovials |
| Crystal Mount-Base-Assembly HTB-M2-XX-BY-R Dual Thickness MicroMount Assembly with specified aperture size (xx) on 18mm/SPINE length rods adhered into specified barcoded reusable bases (By) without cryovials |
Crystal Mount-Base-Assembly HTB-M3-XX-BY-R MicroMesh Assembly with specified aperture size (xx) on 18mm/SPINE length rods adhered into specified barcoded reusable bases (By) without cryovials |
Crystal Mount-Base-Assembly HTB-M5-XX-BY-R MicroLoop Assembly with specified aperture size (xx) on 18mm/SPINE length rods adhered into specified barcoded reusable bases (By) without cryovials |
Crystal Mount-Base-Assembly HTB-M5-XXLD-BY-R MicroLoop LD Assembly with specified aperture size (xx) on 18mm/SPINE length rods adhered into specified barcoded reusable bases (By) without cryovials |
Crystal Mount-Base-Assembly HTB-M8-XX-BY-R MicroLoop E Assembly with specified aperture size (xx) on 18mm/SPINE length rods adhered into specified barcoded reusable bases (By) without cryovials |
Crystal Mount-Base-Assembly HTB-M7-XX-BY-R MicroGripper Assembly with specified aperture size (xx) on 18mm/SPINE length rods adhered into specified barcoded reusable bases (By) without cryovials |
| Crystal Mount-Base-Vial-Assembly A-M2-XX-BY Dual Thickness MicroMount Assembly with specified aperture size (xx) on 18mm/SPINE length rods adhered into specified bases (By) with cryovials |
Crystal Mount-Base-Vial-Assembly A-M3-XX-BY MicroMesh Assembly with specified aperture size (xx) on 18mm/SPINE length rods adhered into specified bases (By) with cryovials |
Crystal Mount-Base-Vial-Assembly A-M5-XX-BY MicroLoop Assembly with specified aperture size (xx) on 18mm/SPINE length rods adhered into specified bases (By) with cryovials |
Crystal Mount-Base-Vial-Assembly A-M5-XXLD-BY MicroLoop LD Assembly with specified aperture size (xx) on 18mm/ SPINE length rods adhered into specified bases (By) with cryovials |
Crystal Mount-Base-Vial-Assembly A-M8-XX-BY MicroLoop E Assembly with specified aperture size (xx) on 18mm/SPINE length rods adhered into specified bases (By) with cryovials |
Crystal Mount-Base-Vial-Assembly A-M7-XX-BY MicroGripper Assembly with specified aperture size (xx) on 18mm/SPINE length rods adhered into specified bases (By) with cryovials |
| Crystal Mount-Base-Vial-Assembly A-M2-XX-BY-R Dual Thickness MicroMount Assembly with specified aperture size (xx) on 18mm/SPINE length rods adhered into specified reusable bases (By) with cryovials |
Crystal Mount-Base-Vial-Assembly A-M3-XX-BY-R MicroMesh Assembly with specified aperture size (xx) on 18mm/SPINE length rods adhered into specified reusable bases (By) with cryovials |
Crystal Mount-Base-Vial-Assembly A-M5-XX-BY-R MicroLoop Assembly with specified aperture size (xx) on 18mm/SPINE length rods adhered into specified reusable bases (By) with cryovials |
Crystal Mount-Base-Vial-Assembly A-M5-XXLD-BY-R MicroLoop LD Assembly with specified aperture size (xx) on 18mm/ SPINE length rods adhered into specified reusable bases (By) with cryovials |
Crystal Mount-Base-Vial-Assembly A-M8-XX-BY-R MicroLoop E Assembly with specified aperture size (xx) on 18mm/SPINE length rods adhered into specified reusable bases (By) with cryovials |
Crystal Mount-Base-Vial-Assembly A-M7-XX-BY-R MicroGripper Assembly with specified aperture size (xx) on 18mm/SPINE length rods adhered into specified reusable bases (By) with cryovials |
| Crystal Mount-Base-Assembly B-M2-XX-BY Dual Thickness MicroMount Assembly with specified aperture size (xx) on 18mm/SPINE length rods adhered into specified bases (By) without cryovials |
Crystal Mount-Base-Assembly B-M3-XX-BY MicroMesh Assembly with specified aperture size (xx) on 18mm/SPINE length rods adhered into specified bases (By) without cryovials |
Crystal Mount-Base-Assembly B-M5-XX-BY MicroLoop Assembly with specified aperture size (xx) on 18mm/SPINE length rods adhered into specified bases (By) without cryovials |
Crystal Mount-Base-Assembly B-M5-XXLD-BY MicroLoop LD Assembly with specified aperture size (xx) on 18mm/SPINE length rods adhered into specified bases (By) without cryovials |
Crystal Mount-Base-Assembly B-M8-XX-BY MicroLoop E Assembly with specified aperture size (xx) on 18mm/SPINE length rods adhered into specified bases (By) without cryovials |
Crystal Mount-Base-Assembly B-M7-XX-BY MicroGripper Assembly with specified aperture size (xx) on 18mm/SPINE length rods adhered into specified bases (By) without cryovials |
| Crystal Mount-Base-Assembly B-M2-XX-BY-R Dual Thickness MicroMount Assembly with specified aperture size (xx) on 18mm/SPINE length rods adhered into specified reusable bases (By) without cryovials |
Crystal Mount-Base-Assembly B-M3-XX-BY-R MicroMesh Assembly with specified aperture size (xx) on 18mm/SPINE length rods adhered into specified reusable bases (By) without cryovials |
Crystal Mount-Base-Assembly B-M5-XX-BY-R MicroLoop Assembly with specified aperture size (xx) on 18mm/SPINE length rods adhered into specified reusable bases (By) without cryovials |
Crystal Mount-Base-Assembly B-M5-XXLD-BY-R MicroLoop LD Assembly with specified aperture size (xx) on 18mm/SPINE length rods adhered into specified reusable bases (By) without cryovials |
Crystal Mount-Base-Assembly B-M8-XX-BY-R MicroLoop E Assembly with specified aperture size (xx) on 18mm/SPINE length rods adhered into specified reusable bases (By) without cryovials |
Crystal Mount-Base-Assembly B-M7-XX-BY-R MicroGripper Assembly with specified aperture size (xx) on 18mm/SPINE length rods adhered into specified reusable bases (By) without cryovials |



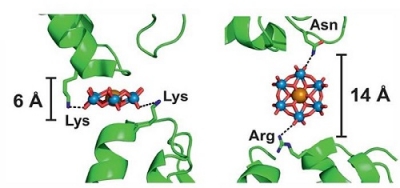



































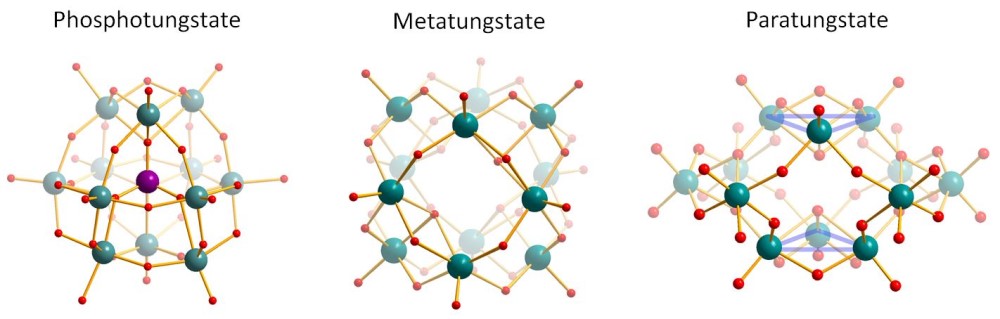 The Tungstate Cluster Kits consist of 6 ready-to-use aliquots of Phosphotungstate, Metatungstate or Paratungstate salts, respectively. All Tungsten clusters contain 12 Tungsten metal centers bridged by Oxygen atoms, but differ in their resulting negative charge (3-, 6- and 10-, respectively).
The Tungstate Cluster Kits consist of 6 ready-to-use aliquots of Phosphotungstate, Metatungstate or Paratungstate salts, respectively. All Tungsten clusters contain 12 Tungsten metal centers bridged by Oxygen atoms, but differ in their resulting negative charge (3-, 6- and 10-, respectively).
 Schematic cross section of a grid hole with ideally distributed single particles in random orientation.
Schematic cross section of a grid hole with ideally distributed single particles in random orientation.