To meet your research needs, Jena Bioscience offers more than 1000 functionally active and highly purified recombinant proteins. Our portfolio covers a wide range of functions and applications – see below for details.
Proteins
Recombinant Proteins
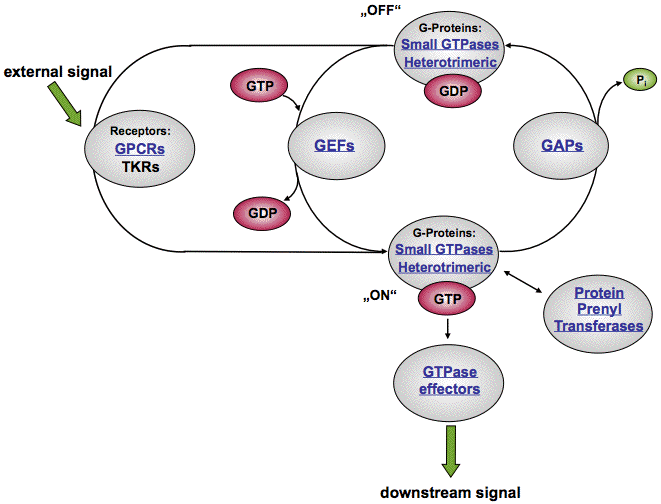
The term GTPase, GTP-binding protein – or simply G-protein – is used for enzymes that are involved in cellular signal transduction by utilizing the nucleotide GTP. They consist of 2 major subgroups called *Heterotrimeric G-proteins* and *Small GTPases*, and their activities are modulated by GEFs and GAPs in a cycling fashion (see figure below).
Traditionally, measuring the activation state of a small GTPase in vivo (i.e. the GTPase-GDP/GTP levels) required tedious 32P-labelling of cells, enzyme isolation and HPLC of bound GDP and GTP.
All antibodies are recommended for Western Blot, Immunohistochemistry, Immunofluoerescence, Immunoprecipitation (IP) and ELISA.
Simplified Illustration of GTP Signal Transduction
Small GTPases (Ras Superfamily)
Dendrogram of the Ras superfamily
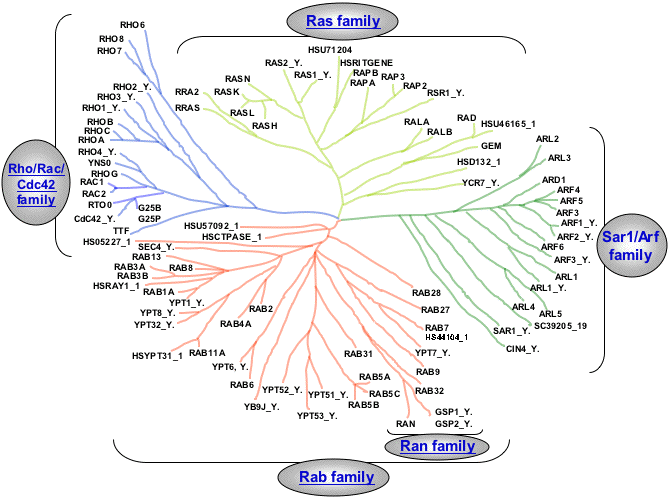
(taken from: Takai et al. (2001) Small GTP-Binding Proteins. Physiol. Rev. 81 (1):153.)
GTPases of the Ras superfamily represent a group of more than 100 known proteins involved in the control of numerous essential cellular functions, and in particular cell proliferation. They were grouped, according to sequence homologies, into five principal branches: Ras family, Rab family, Ran family, Rho/Rac/Cdc42 family and Sar1/Arf family.
Mutations in Ras proteins that block them in a constitutively active state are found in 30 to 60% of human malignancies. Ras-proteins have a molecular mass of 21 kDa. Suitable as a substrate for farnesyltransferase. While mutations in Ras proteins are found in 30 to 60% of human malignancies, other GTPases like Rho and Rac have also been linked to oncogenesis.
Rab proteins are small GTP-binding proteins that form the largest family within the Ras superfamily. Rab proteins regulate vesicular trafficking pathways, behaving as membrane-associated molecular switches.
Ran (Ras-related nuclear protein) is involved in diverse cellular processes like nucleo-cytosplasmic transport of proteins into the nucleus and RNA into the cytoplasm, mitotic spindle assembly, and post-mitotic nuclear assembly. The Ran-regulated nucleo-cytoplasmic transport through the nuclear core complex occurs in conjunction with importins and exportins.
Small GTPases: Ras family
| Products & Ordering | |
| H-Ras PR-107 Harvey rat sarcoma viral oncogene homolog human, recombinant, E. coli | H-RasGST PR-364 Harvey rat sarcoma viral oncogene homolog human, recombinant, E. coli |
| H-Ras ΔC (S17N) PR-202 Harvey rat sarcoma viral oncogene homolog, residues 1-166 human, recombinant, E. coli | H-Ras (Q61L) PR-203 Harvey rat sarcoma viral oncogene homolog human, recombinant, E. coli |
| H-Ras (G12V) PR-206 Harvey rat sarcoma viral oncogene homolog human, recombinant, E. coli | H-Ras (G12V)GST PR-358 Harvey rat sarcoma viral oncogene homolog human, recombinant, E. coli |
| TC21/R-Ras2GST PR-359 Teratocarcinoma oncogene, related ras viral oncogene homolog 2 human, recombinant, E. coli | Rap1B ΔC PR-301 Ras-proximate 1B, deletion of 17 C-terminal residues human, recombinant, E. coli |
| Rheb PR-126 Ras-related GTPase, GST-fusion human, recombinant, E. coli | KRasHis – G-domain PR-239 Kirsten rat sarcoma viral oncogene homolog, G-domain human, recombinant, E. coli |
Small GTPases: Rab family
| Products & Ordering | |||
| Rab1AGST-His PR-113 Ras-associated, small GTP-binding protein mouse, recombinant, E. coli | Rab1BGST-His PR-114 Ras-associated, small GTP-binding protein rat, recombinant, E. coli | Rab2GST-His PR-112 Ras-associated, small GTP-binding protein human, recombinant, E. coli | Rab3AGST-His PR-115 Ras-associated, small GTP-binding protein mouse, recombinant, E. coli |
| Rab4GST-His PR-116 Ras-associated, small GTP-binding protein human, recombinant, E. coli | Rab4BGST-His PR-183 Ras-associated, small GTP-binding protein human, recombinant, E. coli | Rab5A PR-184 Ras-associated, small GTP-binding protein rat, recombinant, E. coli | Rab6A PR-109 Member of Ras oncogene family human, recombinant, E. coli |
| Rab7 PR-108 Ras-associated, small GTP-binding protein dog, recombinant, E. coli | Rab7GST-His PR-362 Ras-associated, small GTP-binding protein dog, recombinant, E. coli | Rab9GST-His PR-185 Ras-associated, small GTP-binding protein human, recombinant, E. coli | Rab14 PR-187 Ras-associated, small GTP-binding protein rat, recombinant, E. coli |
| Rab17GST-His PR-118 Ras-associated, small GTP-binding protein mouse, recombinant, E. coli | Rab18AGST-His PR-188 Ras-associated, small GTP-binding protein mouse, recombinant, E. coli | Rab19GST-His PR-189 Ras-associated, small GTP-binding protein mouse, recombinant, E. coli | Rab22AGST-His PR-190 Ras-associated, small GTP-binding protein human, recombinant, E. coli |
| Rab23 PR-191 Ras-associated, small GTP-binding protein mouse, recombinant, E. coli | Rab24GST-His PR-120 Ras-associated, small GTP-binding protein mouse, recombinant, E. coli | Rab27AGST-His PR-122 Ras-associated, small GTP-binding protein rat, recombinant, E. coli | Rab27 delta C26 PR-192 Ras-associated, small GTP-binding protein rat, recombinant, E. coli |
| Rab31GST-His PR-193 Ras-associated, small GTP-binding protein human, recombinant, E. coli | Rab32 PR-194 Ras-associated, small GTP-binding protein human, recombinant, E. coli | Rab37GST-His PR-196 Ras-associated, small GTP-binding protein human, recombinant, E. coli | RabGDI PR-110 Rab GDP Dissociation Inhibitor, member of Ras oncogene family bovine, recombinant, Sf9 insect cells |
Small GTPases: Ran family
Small GTPases: Rho/Rac/Cdc42 Family
| Products & Ordering | |
| Cdc42 ΔCGST PR-302 Cell Division Cycle Protein 42, C-terminal deletion of 13 residues human, recombinant, E. coli | Cdc42 ΔC G12VGST PR-300 Cell Division Cycle Protein 42, G12V mutant, C-terminal deletion of 13 residues human, recombinant, E. coli |
| Rac1 ΔC PR-303 Ras-related C3 botulinum toxin substrate 1, residues 1-187 human, recombinant, E. coli | RhoA PR-106 Ras-like GTP-binding protein human, recombinant, E. coli |
| RhoAGST PR-363 Ras-like GTP-binding protein, GST-tagged human, recombinant, E. coli |
GEFs (Guanine Nucleotide Exchange Factors)
Small GTPases are normally membrane associated independently of their activation state. Interaction with a specific “guanidine exchange factor” (GEF) induces the GDP – GTP exchange and thereby activates the GTPase, allows for interaction with effector proteins, and localizes and/or activates these proteins.
| Products & Ordering |
| RCC1 (RanGEF) PR-306 Regulator of Chromosome Condensation, Ran Guanine Exchange Factor human, recombinant, E. coli |
| RalGDS-RBDGST PR-365 Ras Binding Domain of the Ral Guanine Nucleotide Dissociation Stimulator human, recombinant, E. coli, Recognized GTPases: Rap-1A, Rap-1B, Rap-2A, Rap-2B, Rit, Rin |
GTPase Effectors
GDP-bound inactive G-proteins can be opened up by guanine nucleotide exchange factors (GEFs) causing the dissociation of GDP and the subsequent binding of GTP. This switches “on” the GTP signal transduction pathway alllowing the binding and activation of downstream effectors like ion channels or enzymes including adenylyl cyclases, kinases → Molecular Biology, and phosphatases → Molecular Biology.
| Products & Ordering | |
| RafRBD (c-Raf) PR-305 Ras Binding Domain of Raf, c-Raf 1 Kinase human, recombinant, E. coli | RafRBDGST (C-Raf) PR-366 Ras Binding Domain of Raf, C-Raf 1 Kinase human, recombinant, E. coli |
| RalGDS-RBDGST PR-365 Ras Binding Domain of the Ral Guanine Nucleotide Dissociation Stimulator human, recombinant, E. coli, Recognized GTPases: Rap-1A, Rap-1B, Rap-2A, Rap-2B, Rit, Rin | RhotekinRBDGST PR-367 Rhotekin Ras Binding Domain, from the Japanese human, recombinant, E. coli |
| WASP-121GST PR-369 Wiskott-Aldrich Syndrome Protein human, recombinant, E. coli, Recognized GTPases: Cdc42, TC10,TCL | AF6-RBDGST (residues 1-141) PR-398 GTP-H-Ras binding cytoplasmic protein, Afadin – Ras-binding domain human, recombinant, E. coli |
| PAK1B-CRIB GST PR-399 P21 activated kinase 1, Cdc42/Rac interactive binding region (AA 56-141), GST-tagged human, recombinant, E. coli |
Protein Prenyl Transferases
Please check our Protein Prenylation Section for engineered protein prenyl transferase mutants, innovative lipid donors, substrates and selective inhibitors
| Products & Ordering | |
| GGTase-I PR-101 Protein geranylgeranyltransferase type I rat, recombinant, E. coli | GGTase-IGST PR-360 Protein geranylgeranyltransferase type I rat, recombinant, E. coli |
| GGTase-II (RabGGTase) PR-103 Protein geranylgeranyltransferase type II, α- and β-subunit rat, recombinant, E. coli | FTase PR-102 Protein farnesyltransferase, α- and β-subunit rat, recombinant, E. coli |
| REP-1 His PR-105 Rab Escort Protein fish, recombinant, E. coli | REP-2His PR-104 Rab Escort Protein human, recombinant, Saccharomyces cerevisiae |
GTPase Activity Assay
| Products & Ordering | |
| RafRBD (c-Raf) PR-305 Ras Binding Domain of Raf, c-Raf 1 Kinase human, recombinant, E. coli | RafRBDGST (C-Raf) PR-366 Ras Binding Domain of Raf, C-Raf 1 Kinase human, recombinant, E. coli |
| RalGDS-RBDGST PR-365 Ras Binding Domain of the Ral Guanine Nucleotide Dissociation Stimulator human, recombinant, E. coli, Recognized GTPases: Rap-1A, Rap-1B, Rap-2A, Rap-2B, Rit, Rin | WASP-121GST PR-369 Wiskott-Aldrich Syndrome Protein human, recombinant, E. coli, Recognized GTPases: Cdc42, TC10,TCL |
Jena Bioscience offers a broad range of recombinant viral, bacterial and parasitic proteins
- expressed in E. coli, S. cerevisae, P. pastoris or insect cells
- with a purity of >90% (confirmed by SDS-PAGE analysis)
- suitable as antigens in a variety of immunoassays as indicated on the corresponding data sheets
Native Proteins
Due to their unmatched sensitivity, pathogen specific native antigens are widely used for immunodiagnostic assays such as ELISA.
Jena Bioscience offers a broad range of high-quality native viral, bacterial, fungal and parasitic antigens which are
- suitable for both antibody detection or as a positive control in a variety of immunoassays
- functionally tested by ELISA
- inactivated and tested for infectivity prior to release
- available in bulk amounts
| Products & Ordering | |
| Measles Virus Antigen (Premium) PR-BA102-S Strain Edmonston native, Vero cell culture | Measles Virus Antigen PR-BA102 Strain Edmonston native, Vero cell culture |
| Mumps/Parotitis Virus Antigen PR-BA103 Strain Enders native, Vero cell culture | Varicella Zoster Virus (VZV) Antigen PR-BA104 Strain Ellen native, HEL-299 cell culture |
| Varicella Zoster Virus (VZV) Glycoprotein Antigen PR-BA104VSG Strain Ellen native, HEL-299 cell culture | Cytomegalovirus Antigen PR-BA109 Strain Towne native, HEL-299 cell culture |
| FSME (TBE) Virus Antigen PR-BA112L Strain Moscow B-4 native | FSME (TBE) Virus Antigen (Premium) PR-BA112 Strain Moscow B-4 native, Vero cell culture |
| Respiratory Syncytial Virus (RSV) Antigen PR-BA113 Strain Long native, Vero cell culture | Adenovirus Type 2 Hexon Antigen PR-BA128 Human adenovirus 2, Strain Adenoid 6 native, Hep-2 cell culture |
| Rubella Virus Antigen PR-BA129G2 Strain Judith native, Vero cell culture | Coxsackievirus B1 Antigen PR-BA134B1 Strain Conn-5 native, Vero cell culture |
| Coxsackievirus B5 Antigen PR-BA134B5 Strain Faulkner native, Vero cell culture | Coxsackievirus B6 Antigen PR-BA134B6 Strain Schmitt native, Vero cell culture |
| Herpes Simplex Virus (HSV) Type 1 Antigen PR-BA1051 Strain F native, Vero cell culture | Herpes Simplex Virus (HSV) Type 2 Antigen PR-BA1052 Strain G native, Vero cell culture |
| Rotavirus Antigen PR-BA1193 Strain SA11 (Simian rotavirus) native, MA-104 cell culture | Parainfluenza Virus Type 1 Antigen PR-BA1261 Strain Sendai native, MDCK cell culture |
| Parainfluenza Virus Type 2 Antigen PR-BA1262 Strain Greer native, Vero cell culture | Parainfluenza Virus Type 3 Antigen PR-BA1263 Strain C243 native, Vero cell culture |
| Products & Ordering | |
| Borrelia afzelii Antigen PR-BA121 native, Broth suspension | Borrelia garinii Antigen PR-BA121GVS native, Broth suspension |
| Brucella abortus Antigen PR-BA116 S99 – Weybridge Fermentation | Campylobacter jejuni Antigen PR-BA139 native, Blood agar |
| Helicobacter pylori Antigen PR-BA118 native, Solid medium | Legionella pneumophila Antigen PR-BA106 Pool of Legionella pneumophila serovars 1A, 1B, 2, 3, 4, 5 ,6 and 7 native, Solid medium |
| Leptospira biflexa Antigen PR-BA125 Patoc native, Broth culture | Mycoplasma pneumoniae Antigen PR-BA127 Strain FH native, Broth culture |
| Yersinia enterocolitica O:3 Antigen PR-BA138 native, liquid culture | Yersinia enterocolitica O:8 Antigen PR-BA13808 native, liquid culture |
| Yersinia enterocolitica O:9 Antigen PR-BA13809 native, liquid culture | Chlamydia pneumoniae Antigen PR-BA1371 native, BGM cell culture |
| Chlamydia pneumoniae Antigen (Premium) PR-BA1371-S Strain TW-183 native, BGM cell culture | Chlamydia trachomatis PR-BA1372 EB & RB native, Vero cell culture |
| Chlamydia abortus PR-BA1373 EB & RB native, Vero cell culture | B. pertussis Filamentous Hemagglutinin (FHA) PR-BA120F Strain Tohama I native, Broth culture |
| Pertussis Toxin PR-BA120P Strain Tohama I native, Broth culture |
| Products & Ordering |
| Candida albicans Antigen PR-BA117 wild strain native, Solid medium |
| Products & Ordering |
| Toxoplasma gondii Antigen PR-BA110 Strain RH native, Vero cell culture |
Detection & Analysis
Jena Bioscience offers highly specific antibodies suitable for a wide range of immunodetection methods
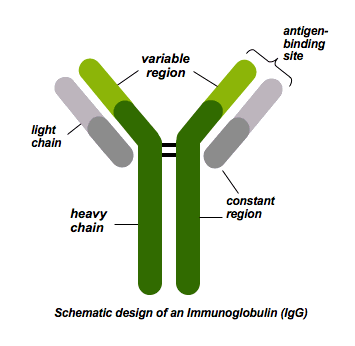
Immunoglobulins are glycoprotein molecules and a group of structurally similar but functionally apparently different proteins. The immunoglobulin “backbone” consists of heavy and light polypeptide chains. Five classes of immunoglobulins (IgA, IgG, IgM, IgE and IgD) have been distinguished on the basis of non cross-reacting antigenic determinants of their heavy chains.
The main function of immunoglobulins is to serve as antibodies that confer humoral immunity against bacterial, viral and other pathogens.
Application of antibodies in research and development, in vitro/in vivo diagnostics, or therapeutics requires antibodies with remarkable specificity.
Antioxidant Antibodies
Antibodies for Methionine Sulfoxide Reductases (MSRs) A and B. MSRs are repair enzymes that protect proteins from oxidative stress by catalyzing stereospecific reduction of oxidized methionine residues.
| Products & Ordering |
| anti-dMSRA ABD-055 anti drosophila Methionine Sulfoxide Reductase A rabbit, polyclonal |
| anti-hMSRA ABD-053 anti human Methionine Sulfoxide Reductase A rabbit, polyclonal |
| anti-hMSRB1 ABD-051 anti human Methionine Sulfoxide Reductase B1 rabbit, polyclonal |
| anti-hMSRB2 ABD-049 anti human Methionine Sulfoxide Reductase B2 rabbit, polyclonal |
| anti-hMSRB3 ABD-050 anti human Methionine Sulfoxide Reductase B3 rabbit, polyclonal |
Neurobiology Antibodies
Neuropeptide and neurotransmitter antibodies derived from immunized rabbits for ELISA and Immunocytochemistry.
| Products & Ordering | |
| anti-PML ABD-030 anti-human PML rabbit, polyclonal | anti-Lom-TK 2 ABD-045 anti-Locusta-Tachykinin 2 rabbit, polyclonal |
| anti-Sp100 ABD-031 anti-human Sp100 rabbit, polyclonal | anti-Head-Peptide ABD-058 rabbit, polyclonal |
| anti-Proctolin ABD-032 rabbit, polyclonal | anti-LMS ABD-059 anti-Leucomyosuppressin rabbit, polyclonal |
| anti-CCAP ABD-033 anti-Crustacean Cardioactive Peptide rabbit, polyclonal | anti-Pam-Igloo-L ABD-060 anti Periplaneta Americana Igloo L 1-75 rabbit, polyclonal |
| anti-PSK ABD-037 anti-Perisulfakinin rabbit, polyclonal | anti-A-AST ABD-062 anti-A-Allatostatin rabbit, polyclonal |
| anti-PSKR ABD-038 anti-Perisulfakinin Receptor rabbit, polyclonal | anti-AKH I ABD-063 anti-Adipokinetic Hormone I rabbit, polyclonal |
| anti-Lom-TK 1 ABD-044 anti-Locusta-Tachykinin 1 rabbit, polyclonal | anti-PVK ABD-065 anti-Periviscerokinin rabbit, polyclonal |
Protein Prenylation
Protein prenylation ranks among the most common lipid modifications of proteins, affecting up to 2% of the proteins in the mammalian proteome.
It involves the formation of a thioether linkage between a 15-carbon or 20-carbon isoprenoid moiety and one or two cysteine residues near the C-terminus of the target protein. Farnesylated and geranylgeranylated proteins include members of the Ras, Rho, Rac, Rap and Rab families, the γ subunit of heterotrimeric G proteins, centromeric proteins and many more.
The prenyl moiety is essential for the reversible association of the target protein with the intracellular membrane and modulates protein-protein interactions.
Jena Bioscience’s innovative protein prenylation products provide the basis for next generation prenylome studies with important implications for human health and disease.
Protein Prenyl Transferases
FTase and GGTase-I are designated CaaX prenyltransferases, since they recognize their protein substrates by the so-called CaaX-box. Both enzymes share a common α subunit (48 kDa) and a homologous β subunit (46 kDa in FTase, 42 kDa in GGTase-I). GGTase-II or RabGGTase differs from FTase and GGTase-I, both structurally and functionally. The enzyme attaches geranylgeranyl groups to two C-terminal cysteines of Rab proteins that terminate in CC or CXC motifs.
Engineered mutants of FTase (Cat.# PR-952 and PR-953) and GGTase-I (Cat.# PR-954) in combination with our innovative and universal lipid donor B-GPP (Cat.# LI-015) allow the detection of femtomolar amounts of prenylatable proteins in eukaryotic cells and tissues and thereby offer a new generation of prenylation studies.
| Products & Ordering | |
| FTase PR-102 Protein farnesyltransferase, α- and β-subunit rat, recombinant, E. coli | FTaseW102T_Y154TGST PR-952 Protein farnesyltransferase mutant, α- and β-subunit rat, recombinant, E. coli |
| GGTase-I PR-101 Protein geranylgeranyltransferase type I rat, recombinant, E. coli | GGTase-IGST PR-360 Protein geranylgeranyltransferase type I rat, recombinant, E. coli |
| GGTase-II (RabGGTase) PR-103 Protein geranylgeranyltransferase type II, α- and β-subunit rat, recombinant, E. coli | REP-1 His PR-105 Rab Escort Protein fish, recombinant, E. coli |
| REP-2His PR-104 Rab Escort Protein human, recombinant, Saccharomyces cerevisiae |
Selected References
Hougland et al. (2009) Getting a handle on protein prenylation. Nat. Chem. Biol. 5 (4):197
Nguyen et al. (2009) Analysis of the eukaryotic prenylome by isoprenoid affinity tagging. Nat. Chem. Biol. 5 (4):227
Lane et al. (2006) Structural biology of protein farnesyltransferase and geranylgeranyltransferase type I. J. Lipid Res. 47:681
Casey et al. (1996) Protein Prenyltransferases. J. Biol. Chem. 271 (10):5289
Protein Prenylation: Lipid Donors
Innovative lipid donors have been developed for sensitive prenylation assays. NBD-FPP and NBD-GPP are a well-established fluorescent analogs of the natural lipid donors.
B-GPP is the first universal lipid donor for all prenylatable proteins, providing versatile prenylation assays with femtomolar detection sensitivity.
| Products & Ordering |
| B-GPP LI-015 Biotin-labeled geranyl pyrophosphate, triethylammonium salt |
| NBD-FPP LI-013 {3,7,11-trimethyl-12-(7-nitro-benzo[1,2,5]oxadiazo-4-ylamino), -dodeca-2,6,10-trien-1} pyrophosphate Fluorescent lipid donor for protein geranylgeranyltransferases (RabGGTase, GGTase-I) |
| NBD-GPP LI-014 3,7-dimethyl-8-(7-nitro-benzo[1,2,5]oxadiazol-4-ylamino), -octa-2,6-diene-1- pyrophosphate Fluorescent lipid donor for protein farnesyltransferase (FTase) |
Selected References
Nguyen et al. (2009) Analysis of the eukaryotic prenylome by isoprenoid affinity tagging. Nature Chemical Biology. 5 (4):227
Wu et al. (2007) Synthesis of a fluorescent analogue of geranylgeranyl pyrophosphate and its use in a high-throughput fluorometric assay for Rab geranylgeranyltransferase. Nat. Protoc. 2 (11):2704
Dursina et al. (2006) Identification and specificity profiling of protein prenyltransferase inhibitors using new fluorescent phosphoisoprenoids. J. Am. Chem. Soc. 128 (9):2822
FTase Substrates and Related Proteins
Since the initial discovery of farnesylated fungal proteins, many more proteins have been demonstrated to contain a prenyl modification. Today it’s well-known, that protein prenylation affects up to 2% of the proteins in the mammalian proteome.
The proteins listed below are FTase substrates and corresponding proteins.
A farnesyl moiety is attached to the cysteine of the so called CaaX box, a C-terminal motif required for farnesylation.
Please note that deletion mutants with truncated C-terminus are not recognized by the protein farnesyltransferase and will thus not be farnesylated.
| Products & Ordering | |
| H-Ras PR-107 Harvey rat sarcoma viral oncogene homolog human, recombinant, E. coli | H-RasGST PR-364 Harvey rat sarcoma viral oncogene homolog human, recombinant, E. coli |
| H-Ras ΔC (S17N) PR-202 Harvey rat sarcoma viral oncogene homolog, residues 1-166 human, recombinant, E. coli | H-Ras (Q61L) PR-203 Harvey rat sarcoma viral oncogene homolog human, recombinant, E. coli |
| H-Ras (G12V) PR-206 Harvey rat sarcoma viral oncogene homolog human, recombinant, E. coli | H-Ras (G12V)GST PR-358 Harvey rat sarcoma viral oncogene homolog human, recombinant, E. coli |
| TC21/R-Ras2GST PR-359 Teratocarcinoma oncogene, related ras viral oncogene homolog 2 human, recombinant, E. coli | Rap1B ΔC PR-301 Ras-proximate 1B, deletion of 17 C-terminal residues human, recombinant, E. coli |
| Rheb PR-126 Ras-related GTPase, GST-fusion human, recombinant, E. coli | KRasHis – G-domain PR-239 Kirsten rat sarcoma viral oncogene homolog, G-domain human, recombinant, E. coli |
GGTase-I Substrates and Related Proteins
Since the initial discovery of prenylated fungal proteins, many more proteins have been demonstrated to contain a prenyl modification. Today it’s well-known, that protein prenylation affects up to 2% of the proteins in the mammalian proteome.
The proteins listed below are GGTase-I substrates and corresponding proteins.
A geranylgeranyl moiety is attached to the cysteine of the so called CaaX box, a C-terminal motif required for prenylation.
Please note that deletion mutants with truncated C-terminus are not recognized by the protein geranylgeranyltransferase and will thus not be prenylated.
| Products & Ordering | |
| RhoA PR-106 Ras-like GTP-binding protein human, recombinant, E. coli | RhoAGST PR-363 Ras-like GTP-binding protein, GST-tagged human, recombinant, E. coli |
| Cdc42 ΔCGST PR-302 Cell Division Cycle Protein 42, C-terminal deletion of 13 residues human, recombinant, E. coli | Cdc42 ΔC G12VGST PR-300 Cell Division Cycle Protein 42, G12V mutant, C-terminal deletion of 13 residues human, recombinant, E. coli |
| Rac1 ΔC PR-303 Ras-related C3 botulinum toxin substrate 1, residues 1-187 human, recombinant, E. coli | PAK1B-CRIB GST PR-399 P21 activated kinase 1, Cdc42/Rac interactive binding region (AA 56-141), GST-tagged human, recombinant, E. coli |
RabGGTase Substrates and Related Proteins
Since the initial discovery of prenylated fungal proteins, many more proteins have been demonstrated to contain a prenyl modification. Today it’s well-known, that protein prenylation affects up to 2% of the proteins in the mammalian proteome.
The proteins listed below are RabGGTase substrates and corresponding proteins.
A geranylgeranyl moiety is attached to one or two C-terminal cysteines of Rab proteins with a hypervariable C-terminal domain. RabGGTase does not recognize its substrate directly, but exerts its function in concert with Rab Escort Protein (REP).
Please note that deletion mutants with truncated C-terminus are not recognized by the protein geranylgeranyltransferase and will thus not be prenylated.
| Products & Ordering | |
| Rab1AGST-His PR-113 Ras-associated, small GTP-binding protein mouse, recombinant, E. coli | Rab1BGST-His PR-114 Ras-associated, small GTP-binding protein rat, recombinant, E. coli |
| Rab2GST-His PR-112 Ras-associated, small GTP-binding protein human, recombinant, E. coli | Rab3AGST-His PR-115 Ras-associated, small GTP-binding protein mouse, recombinant, E. coli |
| Rab4GST-His PR-116 Ras-associated, small GTP-binding protein human, recombinant, E. coli | Rab4BGST-His PR-183 Ras-associated, small GTP-binding protein human, recombinant, E. coli |
| Rab5A PR-184 Ras-associated, small GTP-binding protein rat, recombinant, E. coli | Rab6A PR-109 Member of Ras oncogene family human, recombinant, E. coli |
| Rab7 PR-108 Ras-associated, small GTP-binding protein dog, recombinant, E. coli | Rab7GST-His PR-362 Ras-associated, small GTP-binding protein dog, recombinant, E. coli |
| Rab9GST-His PR-185 Ras-associated, small GTP-binding protein human, recombinant, E. coli | Rab14 PR-187 Ras-associated, small GTP-binding protein rat, recombinant, E. coli |
| Rab17GST-His PR-118 Ras-associated, small GTP-binding protein mouse, recombinant, E. coli | Rab18AGST-His PR-188 Ras-associated, small GTP-binding protein mouse, recombinant, E. coli |
| Rab19GST-His PR-189 Ras-associated, small GTP-binding protein mouse, recombinant, E. coli | Rab22AGST-His PR-190 Ras-associated, small GTP-binding protein human, recombinant, E. coli |
| Rab23 PR-191 Ras-associated, small GTP-binding protein mouse, recombinant, E. coli | Rab24GST-His PR-120 Ras-associated, small GTP-binding protein mouse, recombinant, E. coli |
| Rab27AGST-His PR-122 Ras-associated, small GTP-binding protein rat, recombinant, E. coli | Rab27 delta C26 PR-192 Ras-associated, small GTP-binding protein rat, recombinant, E. coli |
| Rab31GST-His PR-193 Ras-associated, small GTP-binding protein human, recombinant, E. coli | Rab32 PR-194 Ras-associated, small GTP-binding protein human, recombinant, E. coli |
| Rab37GST-His PR-196 Ras-associated, small GTP-binding protein human, recombinant, E. coli | RabGDI PR-110 Rab GDP Dissociation Inhibitor, member of Ras oncogene family bovine, recombinant, Sf9 insect cells |
The comprehensive identification of ATP-binding proteins like the important class of kinases and the dynamic analysis of nucleotide-protein interactions at the proteomic scale are fundamental for understanding better the regulatory mechanisms of nucleotide-binding proteins[1].
ATP binding activities as well as specific binding sites of proteins can be detected globally in any biological sample or tissue from any species by using Biotin/Desthiobiotin-conjugated acyl-ATP probes (AcATP)[2-6].
The procedure relies on the following mechanism:
(Scheme adapted from Xiao et al.[3])
• AcATP binds to the pocket of ATP binding proteins and places the acyl group in close proximity to conserved lysine residues.
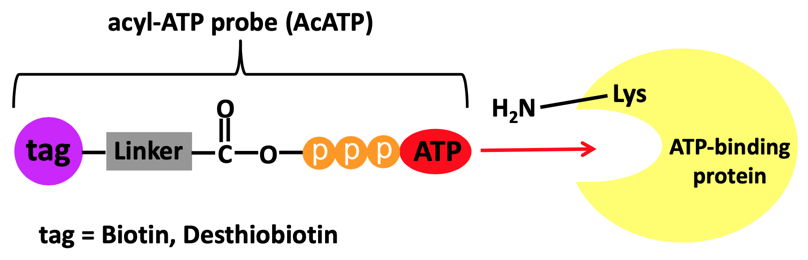
• Nucleophilic attack by the amino group of the lysine at the AcATP probe.
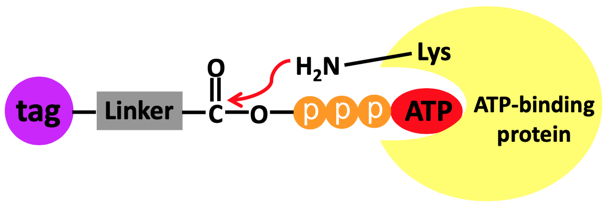
• Amide bond formation resulting in a covalent attachment of the acyl reporter of the AcATP probe on the lysine and a concomitant release of ATP.
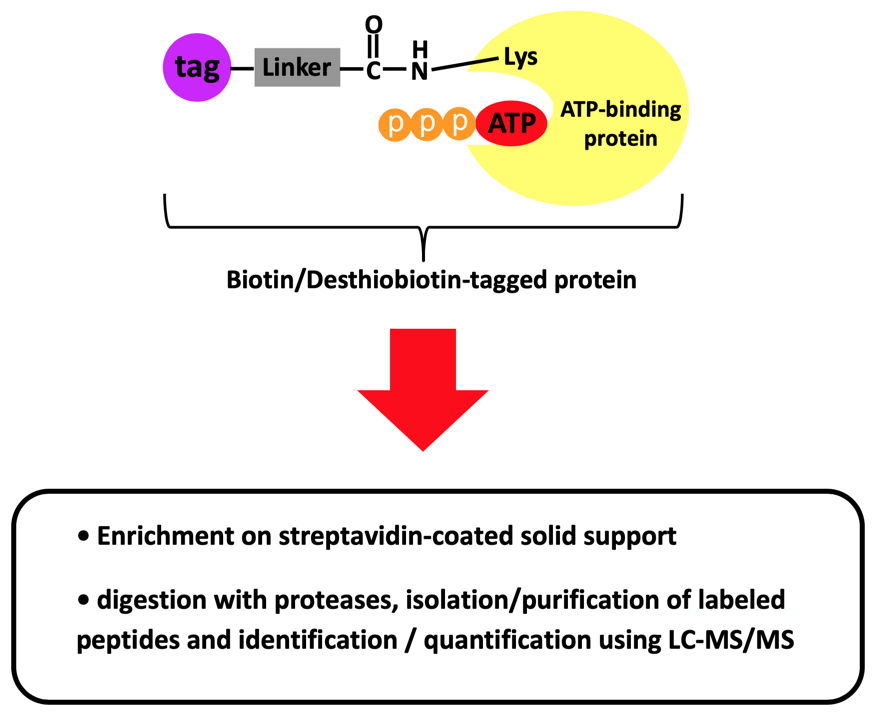
Selected References:
[1] Manning et al. (2002) The protein kinase complement of the human genome. Science 298:1912.
[2] Villamor et al. (2013) Profiling protein kinases and other ATP binding proteins in Arabidopsis using Acyl-ATP probes. Molecular & Cellular Proteomics 12 (9):2481.
[3] Xiao et al. (2013) Proteome-wide discovery and characterizations of nucleotide-binding proteins with affinity-labeled chemical probes. Anal. Chem. 85 (6):3198.
[4] Okerberg et al. (2019) Chemoproteomics using nucleotide acyl phosphates reveals an ATP binding site at the dimer interface of procaspase-6. Biochemistry 58 (52):5320.
[5] Nordin et al. (2015) ATP acyl phosphate reactivity reveals native conformations of Hsp90 paralogs and inhibitor target engagement. Biochemistry 54 (19):3024.
[6] Adachi et al. (2014) Proteome-wide discovery of unknown ATP-binding proteins and kinase inhibitor target proteins using an ATP probe. J. Proteome Res. 13 (12):5461.
Purification
The ability to purify recombinant proteins using affinity chromatography has greatly advanced protein research.
In affinity chromatography, the protein of interest is purified by its ability to bind a specific ligand that is immobilized on a chromatographic bead material (matrix).
Crude cell-lysates are incubated with such a matrix under conditions that ensure specific binding of the protein to the immobilized ligand. Other proteins that do not bind the immobilized ligand are washed through. Elution of the bound protein of interest can be achieved by changing the experimental conditions to favour desorption.
Jena Bioscience offers both Agarose and Magnetic Beads as matrix for different purification tasks.
| Purification of | Agarose | Magnetic Beads |
| mRNA cap binding proteins | m7GTP Agarose | |
| GST-tagged proteins | Glutathione Agarose | Glutathione Magnetic Beads |
| His-tagged proteins | Ni-NTA Agarose | Ni-NTA Magnetic Beads |
Glutathione S-transferase (GST) is a 26 kDa protein derived from Schistosoma japonicum. GST enzymes from various sources, both native and recombinantly expressed as fusion to the N-terminus of target proteins, are easily purified in a one-step procedure by affinity chromatography on immobilized glutathione (Glutathione Agarose).
Due to the positive influence of the GST-tag on protein solubility and expression efficiency especially of small proteins, this technique has been widely used to generate proteins for crystallization, molecular immunology studies and studies involving protein-protein and protein-DNA interactions.
However, the comparably large GST-tag might sometimes interfere with these downstream applications. In these cases it is easily removed by protease cleavage e.g. by Factor Xa provided that a specific protease sequence is located between the protein domain and GST.
Glutathione Magnetic Agarose Beads
Suitable for small-scale & automated purification of Glutathione S Transferase (GST)-tagged proteins.
Glutathione Magnetic Agarose Beads consist of a ferrimagnetic core that is coated with 6 % cross-linked Agarose coupled to gluathione via a 12-atom spacer.
| Glutathione Magnetic Agarose characteristics | |
| Matrix | Ferrimagnetic beads coated with 6 % cross-linked Glutathione Agarose |
| Ligand | Glutathione covalently coupled to 6% Agarose via a neutral 12 atom spacer |
| Medium particle diameter | 30 µm (highly homogenous) |
| Protein Binding capacity* | 10 mg/ml beads |
| Recommended pH | between 3 – 12 |
| Chemical stability | Stable to all solutions commonly used during the purification procedure (refer to the datasheet) |
* Tested with Glutathione-S-transferase (GST) (~25 kDa); Binding capacity may vary depending on amino acid sequence, molecular weight and GST-tag location.
| Products & Ordering |
| Glutathione Magnetic Agarose Beads AC-605 |
Glutathione Agarose
Suitable for FPLC, gravity flow and batch purification
4% cross-linked Glutathione agarose is a superior agarose support with covalently coupled glutathione attached via a 12-atom spacer. Its unique properties facilitate the rapid and high-yielding one-step purification of some native Glutathione S Transferase (GST) enzymes and recombinant GST-tagged proteins. Furthermore, Glutathione agarose is suitable for GST-pull down assays.
- Excellent binding capacity based on high surface area to volume ratio
- Notable mechanical and chemical stability
- Reusable without any significant decrease in yield
| Glutathione Agarose characteristics | |||
| Matrix | 4 % agarose | ||
| Linker | 12-atom spacer | ||
| Bead size | 50 – 150 µm | ||
| Binding capacity* | > 8 mg/ml resin | ||
| Max. linear flow rate | 250 cm/h | ||
| pH stability | 2 – 12 | ||
| Chemical stability | stable in 0.1 M acetate pH 4.0, 0.1 M NaOH, 70 % ethanol or 6 M guanidine hydrochloride for two hours at room temperature, not autoclavable | ||
| Recommended flow rates | |||
| bed volume | inner diameter | sample loading | washing/elution |
| 1 ml | 6.6 mm | 0.2 – 1.0 ml/min | 1.0 ml/min |
| 10 ml | 16 mm | 0.5 – 5.0 ml/min | 5.0 ml/min |
| Products & Ordering |
| Glutathione Agarose AC-210 |
Selected References
[1] Bee et al. (2010) Growth and tumor suppressor NORE1A is a regulatory node between Ras signaling and microtubule nucleation. J. Biol. Chem. 285 (21):16258.
Immobilized metal affinity chromatography (IMAC) is most frequently used for the purification of polyhistidine (His-) tagged proteins. This technique is based on the interaction between certain exposed protein residues (preferentially histidines) with transition metal cation Ni2+. Ni2+ itself is immobilized to a cross-linked agarose matrix via a chelating group such as nitrilotriacetic acid (NTA).
Nickel NTA Magnetic Agarose Beads
Suitable for small-scale & automated purification of polyhistidine (His)-tagged proteins.
Nickel-NTA Magnetic Agarose Beads consist of a ferrimagnetic core that is coated with 6 % cross-linked Agarose coupled to Ni2+-ions via a Nitrilotriacetic acid (NTA)-ligand.
The tetradentate NTA-linker generally results in tighter protein binding and reduced metal leaching, compared to the tridentate Iminodiacetic (IDA)-version.
| Ni-NTA Magnetic Agarose characteristics | |
| Matrix | Ferrimagnetic beads coated with 6 % cross-linked Ni-NTA Agarose |
| Ligand | Nickel (Ni2+) via an Nitrilotriacetic acid (NTA)-linker |
| Ligand density | > 12 µmol Ni2+/ml beads |
| Medium particle diameter | 30 µm (highly homogenous) |
| Protein Binding capacity* | 70 mg/ml beads |
| Recommended pH | between 3–12 |
| Chemical stability | Stable to all solutions commonly used during the purification procedure (refer to the datasheet) |
* Tested with His – GFP (~32 kDa); Binding capacity may vary depending on amino acid sequence, molecular weight and His-tag location.
| Products & Ordering |
| Ni-NTA Magnetic Agarose Beads AC-604 |
Nickel NTA Agarose
Suitable for FPLC, gravity flow and batch purification
His-tagged proteins are efficiently purified by a one-step procedure from crude lysates both under denaturing and non-denaturing conditions.
- High capacity
- Minimum metal leaching
| Product characteristics | |
| Matrix | 6 % cross-linked agarose |
| Bead size | 50-170 µm |
| Linker | Nitrilotriacetic acid (NTA) |
| Binding capacity* | > 50 mg/ml gel |
| Recommended flow rate | 1 ml column – 1.0 ml/min 10 ml column – 5.0 ml/min |
| Maximum linear flow rate | 26 cm/h |
| Maximum pressure | 2.8 psi |
| Recommended pH | between 7 – 8 |
| Chemical stability | Stable to all solutions commonly used in gel filtration including reducing agents (see procedure) |
| Products & Ordering |
| Ni-NTA Agarose AC-501 |
Selected References
[1] Porath et al. (1975) Metal chelate affinity chromatography, a new approach to protein fractionation. Nature 258:598.
[2] Gaberc-Porekar et al. (2001) Perspectives of immobilized-metal affinity chromatography. J. Biochem. Biophys. Methods 49:335.
[3] Ueda et al. (2003) Current and prospective applications of metal ion–protein binding. Journal of Chromatography 988:1.
[4] Liesiene et al. (1997) Immobilized metal affinity chromatography of human growth hormone-effect of ligand density. Journal of Chromatography 764:27.
